Pcl3 Polar Or Nonpolar Asking List
The PCl3 is a polar molecule, and the polarity is due to its tetrahedral geometry with a lone pair of electrons on the phosphorus atom. The electronegativity difference between chlorine and phosphorus creates two opposing dipoles with a more negative charge on chlorine and a positive charge on phosphorus results in polar bond formation.

Polar and Nonpolar Molecules
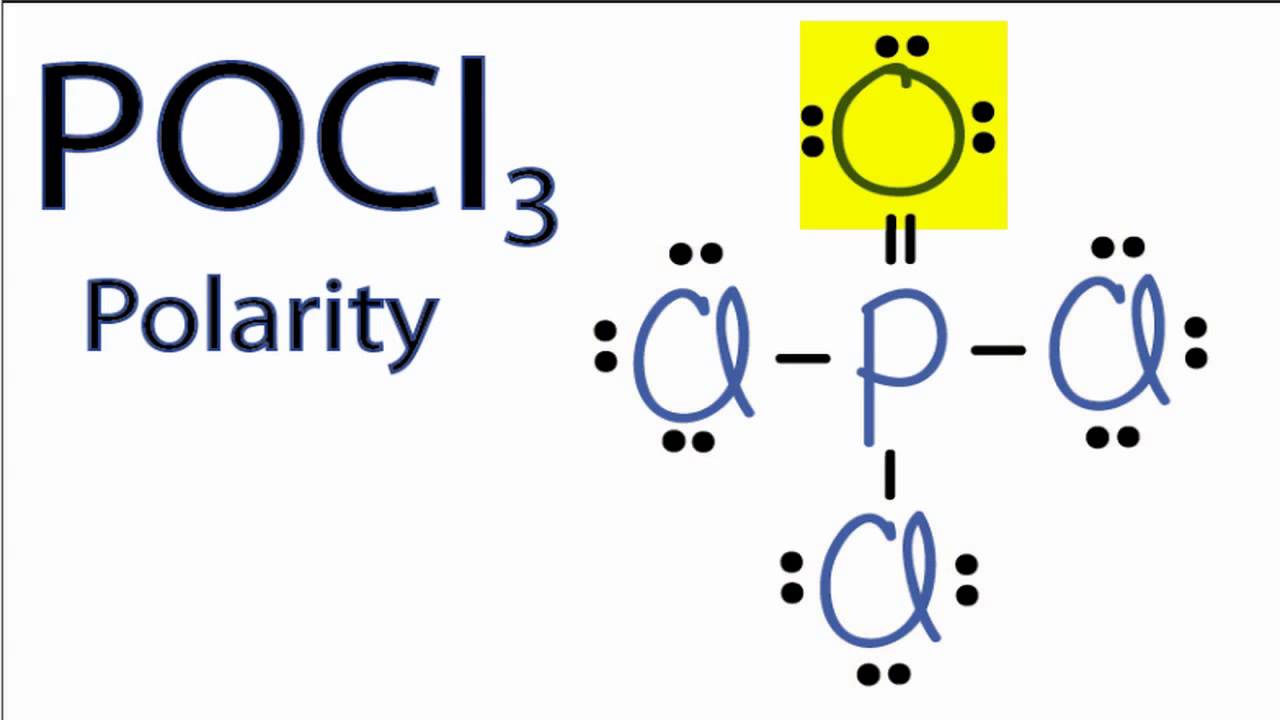
POCl3 is a POLAR molecule because the P=O bond and P-Cl bonds present in the molecule are polar and it has asymmetric geometry which causes the partial positive (ẟ+) and partial negative (ẟ-) charge to appear on the molecule. These ẟ+ and ẟ- charges are responsible to make the entire POCl3 molecule polar.
What Is Pcl3 Lewis Structure?
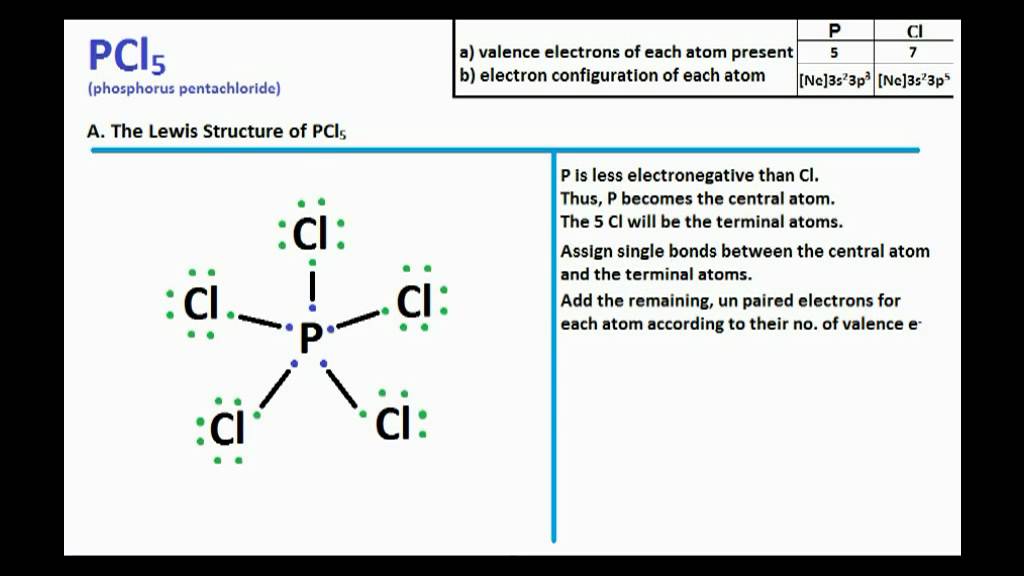
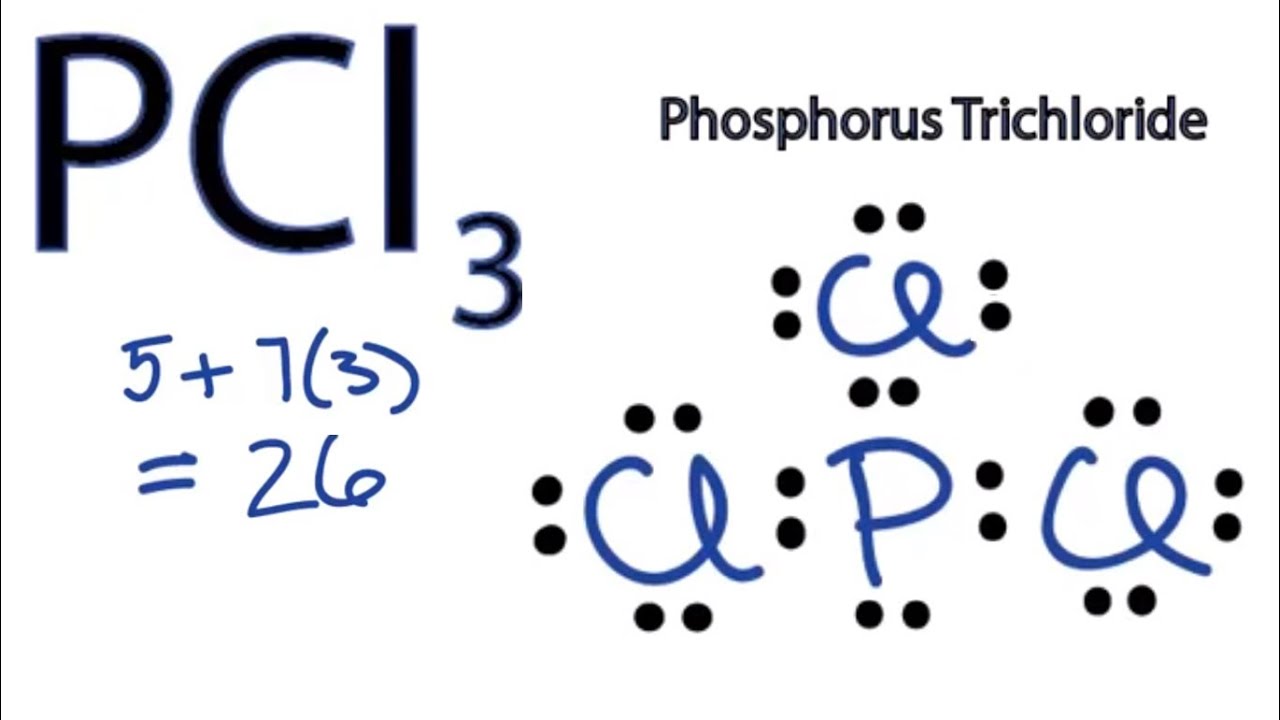
Formula: The chemical formula of phosphorous trichloride is (PCL3). Common Name: The common name of PCL3 is Trichlorophosphane, Phosphorous chloride, and Phosphorus (III) chloride. Table of Contents Is PCL3 Polar Or Nonpolar Summary PCL3 Molecular Geometry Summary PCL3 Lewis Structure Hybridization Of PCL3 Summary Bond Angle Of PCL3 Summary

Pcl3 Polar Or Nonpolar Asking List
Is PCl3 polar or nonpolar? Don't worry, the answer is simple! PCl3 is a polar molecule because of its geometry and difference in electronegativity between the 2 atoms. Again another question like, whether PCl3 is ionic or covalent, can pop up in your mind.
MakeTheBrainHappy Is PCl3 Polar or Nonpolar?
Molecular Polarity. To determine if a molecule is polar or nonpolar, it is frequently useful to look at Lewis structures. Nonpolar compounds will be symmetric, meaning all of the sides around the central atom are identical - bonded to the same element with no unshared pairs of electrons.Polar molecules are asymmetric, either containing lone pairs of electrons on a central atom or having atoms.

How to draw PCl3 Lewis Structure? Science Education and Tutorials
The molecule is symmetric. The two oxygen atoms pull on the electrons by exactly the same amount. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons. Exercise 4.12. 1. Label each of the following as polar or nonpolar.

Is PCl3 (Phosphorus trichloride) Ionic or Covalent/Molecular? YouTube
Phosphoryl Chloride or Phosphorus Oxychloride is a polar molecule due to the uneven distribution of valence electrons in the molecule and a net dipole moment in it. Due to the asymmetry seen in the molecule's shape, the dipole moments are not nullified, making this molecule a polar molecule.

Is PCL3 Polar or Nonpolar? (Phosphorus Trichloride) YouTube
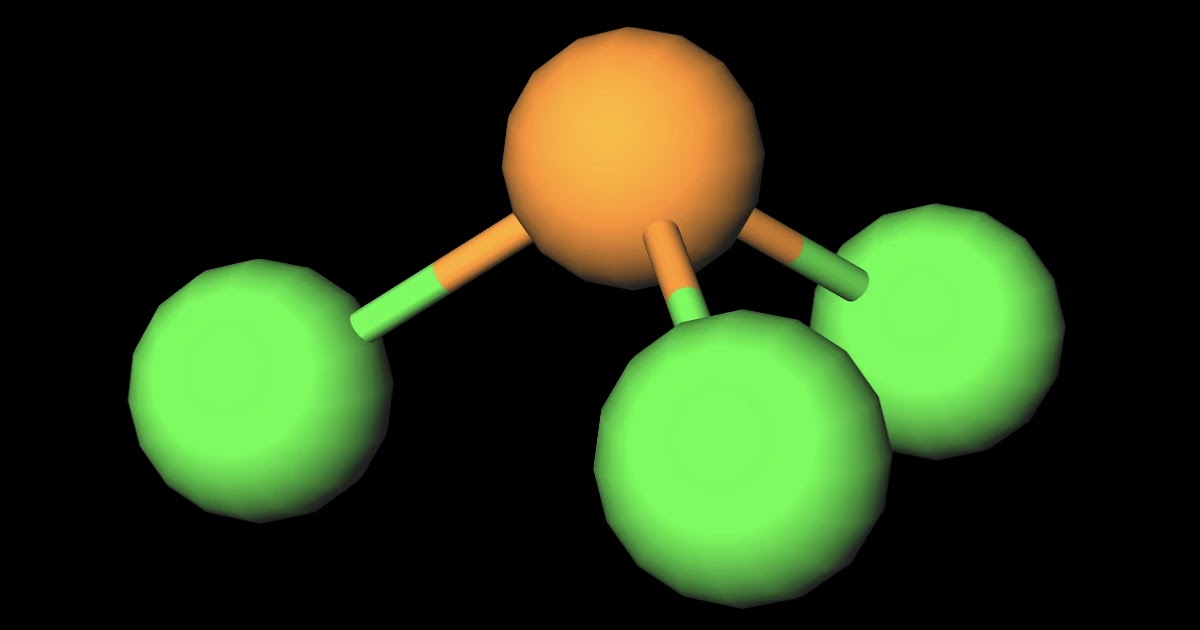
Despite the polar bonds, PCl3 is a nonpolar molecule due to its symmetrical arrangement. PCl3 Lewis Structure Bond Angle. The bond angle in PCl3, or phosphorus trichloride, is a crucial aspect of its molecular geometry. Understanding the bond angle helps us comprehend the overall shape and properties of the molecule.

Is PCl3 (Phosphorous trichloride) Polar or NonPolar YouTube
The bond polarity between two atoms can be estimated if you know the electronegativity of both elements. Calculate the electronegativity difference (ΔEN) and average ( EN) of the two electronegativities, and use the table below to determine the bond type and polarity. Calculate the molecular polarity (polar, non-polar) of a chemical bond based.

Is PCl3 Polar or NonPolar? (Phosphorous trichloride) Yes Dirt
Conclusion FAQ on "Is PCl3 polar or nonpolar?" Is PCl3 a polar compound? What type of bond is PCl3? Is PCl3 a dipole?

PCl3 Lewis Structure, Hybridization, Molecular Geometry, and MO Diagram
PCl3 is a POLAR molecule because the Chlorine (Cl) present in the molecule is more electronegative, which causes the partial positive (ẟ+) and partial negative (ẟ-) charge to appear on the molecule. These ẟ+ and ẟ- charges are responsible to make the entire PCl3 molecule polar.
Is POCl3 Polar or Nonpolar? YouTube
Is PCl3 polar or nonpolar? Question: Is PCl 3 polar or nonpolar? VSEPR: VSEPR is the acronym representing ''valence shell electron pair repulsion.'' It's a theory that utilizes the electron.

PCl3 Lewis Structure and Molecular Geometry YouTube
PCl3 is a polar molecule because of its tetrahedral geometrical shape having a lone pair on Phosphorus atom and the difference between the electronegativity of Chlorine (3.16) and Phosphorus (2.19) atoms resulting in unequal sharing of electrons and develop positive and negative poles across the molecule making it a polar molecule.
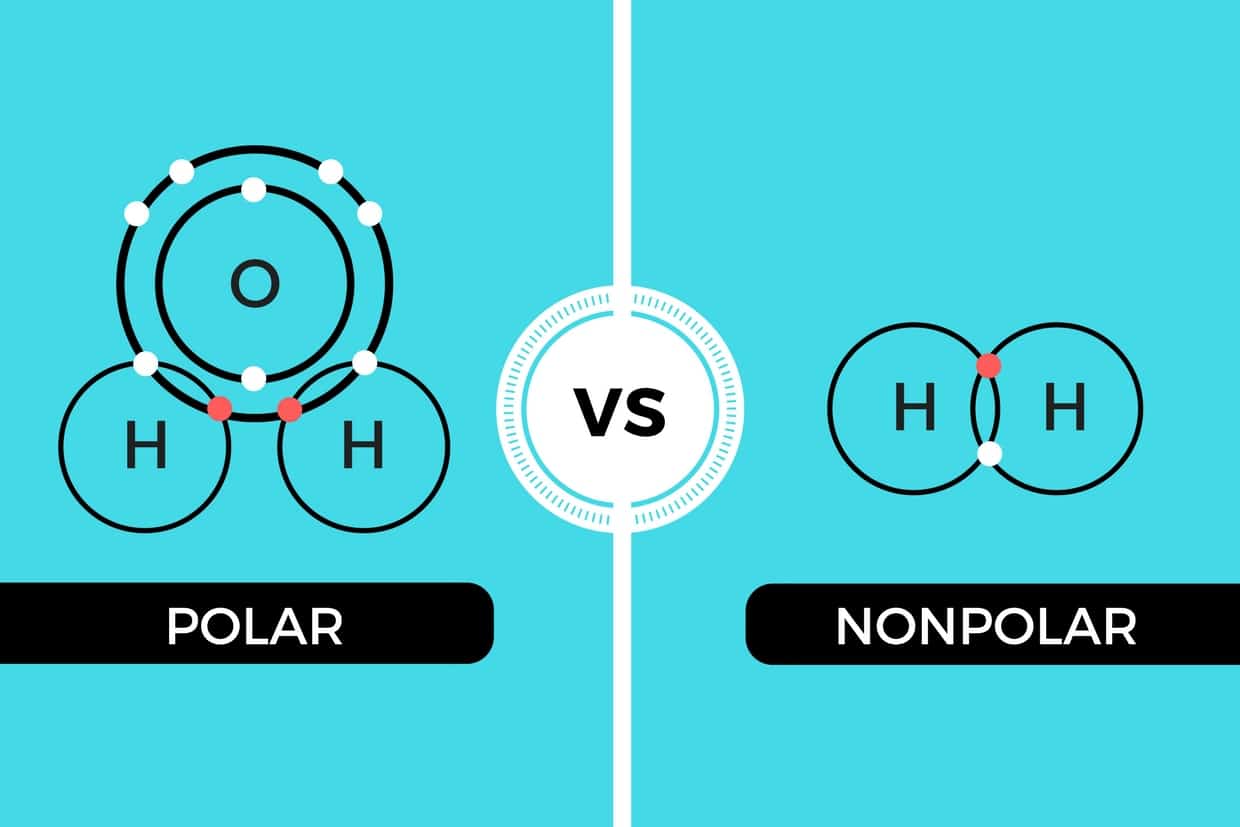
Polar and Nonpolar Covalent Bonds Characteristics & Differences
Phosphorous trichloride molecules are polar because each atom contributes unevenly to the molecule's electron density, making it unequally charged along its three axes.

PCl3 Lewis Structure, Hybridization, Molecular Geometry, and MO Diagram
Wayne Breslyn Join this channel and unlock members-only perks Learn to determine if PCl3 (Phosphorous trichloride) is polar or non-polar based on the Lewis Structure and the molecular.

Is PCl3 Ionic or Covalent? Techiescientist
PCl 3 is a polar molecule because the P-Cl bond is polar, and the three bonds are not equivalent to the lone pair which causes an asymmetrical distribution of bonding electrons in the molecule. This results in a permanent dipole directed towards the P-Cl bonds as drawn: Check this 99-question multiple-choice quiz on Geometry and Hybridization: Free